Being an essential component of pharmaceutical development, stability studies allow the evaluation of product stability under the influence of various environmental conditions. These include temperature, humidity and light, simulating different climatic zones from around the world. The data from such studies can be used by the client to establish recommended storage conditions, retest periods and shelf life.
ALS offer ICH stability storage and testing programmes for a wide range of APIs, pharmaceuticals, biopharmaceuticals, medical devices, chemicals and cosmetics, whether required for initial product registration and/or Product Quality Review (PQR). Our purpose-built walk-in stability rooms are fully validated to meet GMP regulations and can be utilised for both long term and short-term shelf life studies. All rooms are monitored in real time and our monitoring systems are fully validated and compliant with 21 CFR Part 11 requirements.We operate emergency back-up facilities on site, allowing business continuity and complete peace of mind for our clients.
We can provide a solution tailored to your requirements - storage only or full storage plus testing. Samples can be stored and scheduled for testing or shipped to your chosen location at each time point or as required.
Security
- All chambers are kept locked, with restricted access
- Audible and visual alarms for temperature and humidity (above and below set conditions)
- Data loggers have email and auto dial alert functionality
- UPS on data logger ensuring continuous monitoring and alarm call outs

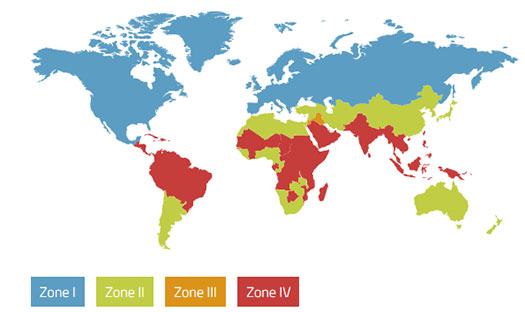
Zone I
Temperate Climate
Zone II
Subtropical and Mediterranean climate
Zone III
Hot and dry climate
Zone IVa
Hot and humid climate
Zone IVb
Hot and very humid climate
Conditions

Stability storage conditions available:
- 5ºC
- Long term conditions for cold stored products or retained/control samples
- 25ºC/60%RH
- Long term conditions for climatic zones I and II
- 30ºC/65%RH
- Intermediate and long term conditions for climatic zones I, II,III and IVa
- 30ºC/75%RH
- Long term conditions for climatic zone IVb
- 40ºC/75%RH
- Accelerated conditions for climatic zones I, II, III and IV
Photostability
ALS also offer photostability testing in accordance with ICHQ1B (option 2).